MALTOTRIOSE, D-(+)-(RG)
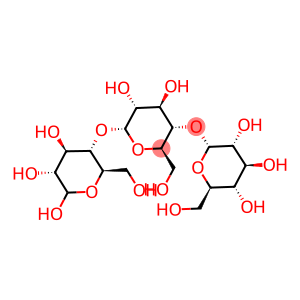
O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranose
CAS: 1109-28-0
Molecular Formula: C18H32O16
MALTOTRIOSE, D-(+)-(RG) - Names and Identifiers
| Name | O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranose |
| Synonyms | Maltotriose MALTOTRIOSE, D-(+)-(RG) 4-O-[4-O-(α-D-Glucopyranosyl)-α-D-glucopyranosyl]-D-glucose (+)-4-O-(4-O-α-D-Glucopyranosyl-α-D-glucopyranosyl)-D-glucose O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranose D-Glucose, O-.alpha.-D-glucopyranosyl-(1?4)-O-.alpha.-D-glucopyranosyl-(1?4)- O-α-DD-Glucopyranosyl-(1-4)-O-α-D-glucopyranosyl-(1-4)-D-glucose, α-D-Glc-(1-4)-α-D-Glc-(1-4)-D-Glc 4-[3,4-dihydroxy-6-(hydroxymethyl)-5-[3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-oxan-2-yl]oxy-2,3,5,6-tetrahydroxy-hexanal |
| CAS | 1109-28-0 |
| EINECS | 214-174-2 |
| InChI | InChI=1/C18H32O16/c19-1-4-7(22)8(23)12(27)17(31-4)34-15-6(3-21)32-18(13(28)10(15)25)33-14-5(2-20)30-16(29)11(26)9(14)24/h4-29H,1-3H2/t4-,5-,6-,7-,8+,9-,10-,11-,12-,13-,14-,15-,16?,17-,18-/m1/s1 |
| InChIKey | FYGDTMLNYKFZSV-DZOUCCHMSA-N |
MALTOTRIOSE, D-(+)-(RG) - Physico-chemical Properties
| Molecular Formula | C18H32O16 |
| Molar Mass | 504.44 |
| Density | 1.4403 (rough estimate) |
| Melting Point | 132-135 °C |
| Boling Point | 513.84°C (rough estimate) |
| Specific Rotation(α) | 160 ° (C=2, H2O) |
| Flash Point | 477°C |
| Water Solubility | Soluble in water. |
| Solubility | Solubility in water: 50 mg/mL, colorless and transparent aqueous solution. |
| Vapor Presure | 6.5E-35mmHg at 25°C |
| Appearance | White powder |
| Color | White to Off-white |
| BRN | 1443481 |
| pKa | 12.39±0.20(Predicted) |
| Storage Condition | Sealed in dry,2-8°C |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents. |
| Refractive Index | 1.6760 (estimate) |
| MDL | MFCD00006629 |
| Physical and Chemical Properties | Hydrolysis of the starch exudate of Saccharomyces cerevisiae. Solubility in water: 50 mg/mL, colorless and transparent aqueous solution. Specific rotation [a]20/D :162.5 °(c = 1, H2O). |
MALTOTRIOSE, D-(+)-(RG) - Risk and Safety
| Safety Description | 24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| FLUKA BRAND F CODES | 10 |
| HS Code | 29400090 |
MALTOTRIOSE, D-(+)-(RG) - Reference Information
| Preparation method | In nature, maltose mainly exists in germinated grains, especially malt, so this name is given. Under the action of starch invertase, starch undergoes a hydrolysis reaction to produce maltoglycan, which undergoes a hydrolysis reaction to produce two molecules of glucose. Maltoglycan is a series of low molecular weight sugars obtained by hydrolysis of starch. It is a linear group of α(1-4) linked D-glucose polymers. Usually maltose industrial grade products are made by acidic hydrolysis of potato starch. Maltotriose (Maltotriose) consists of α(1-4) linked D-glucose monomer condensation with a polymerization degree of 3. |
| main uses | food gelatinization, pigment and brittle agent in food processing. Fabric finishing agents are used to increase the weight and stiffness of fabric fibers. Thickeners and adhesives for pharmaceutical and paper coatings. |
| Biological activity | Maltotriose are common oligosaccharide metabolites, which are often found in human urine after ingestion of maltose. |
| use | used for content determination/identification/pharmacological experiments, etc. |
Last Update:2024-04-09 02:00:06
Supplier List
Multiple SpecificationsSpot supply
Product Name: Maltotriose hydrate Visit Supplier Webpage Request for quotationCAS: 1109-28-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Maltotriose Visit Supplier Webpage Request for quotationCAS: 1109-28-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranose Request for quotation
CAS: 1109-28-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1109-28-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: MALTOTRIOSE Request for quotation
CAS: 1109-28-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1109-28-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple SpecificationsSpot supply
Product Name: Maltotriose Visit Supplier Webpage Request for quotationCAS: 1109-28-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Multiple SpecificationsSpot supply
Product Name: Maltotriose hydrate Visit Supplier Webpage Request for quotationCAS: 1109-28-0
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Spot supply
Product Name: Maltotriose Visit Supplier Webpage Request for quotationCAS: 1109-28-0
Tel: 0714-3999186
Email: 2853786052@qq.com
Mobile: 86+15671228036
QQ: 2853786052
Wechat: 15671228036
Product Name: O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranosyl-(1→4)-O-α-D-glucopyranose Request for quotation
CAS: 1109-28-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1109-28-0
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: MALTOTRIOSE Request for quotation
CAS: 1109-28-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1109-28-0
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Multiple SpecificationsSpot supply
Product Name: Maltotriose Visit Supplier Webpage Request for quotationCAS: 1109-28-0
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History




